|
Each atom may be forming other bonds as well as the one shown - but these are irrelevant to the argument. What happens if two atoms of equal electronegativity bond together?Ĭonsider a bond between two atoms, A and B. Fluorine (the most electronegative element) is assigned a value of 4.0, and values range down to caesium and francium which are the least electronegative at 0.7. The Pauling scale is the most commonly used. 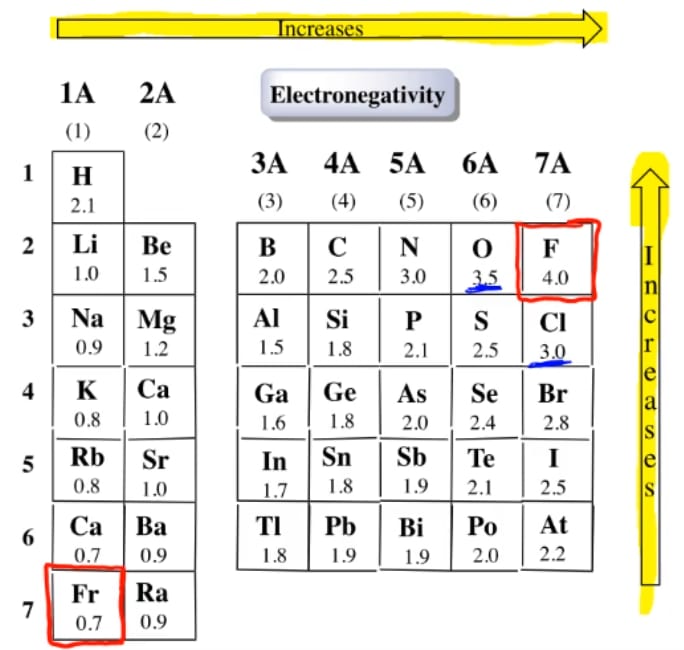
If you are interested in electronegativity in an organic chemistry context, you will find a link at the bottom of this page.Įlectronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. It looks at the way that electronegativity differences affect bond type and explains what is meant by polar bonds and polar molecules. This page explains what electronegativity is, and how and why it varies around the Periodic Table.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
